Molar Mass of a Volatile Liquid LabGroup Members: Steven Soranno, Michael PizzutoPurpose: In this experiment the molar mass of an unknown volatile liquid will be determined by measuring what mass of vapor of the liquid is need to fill a flask of known volume at a particular temperature and pressure.Materials:· 250mLErlenmeyer Flask· 1000mL Beaker· Alumin
...[Show More]
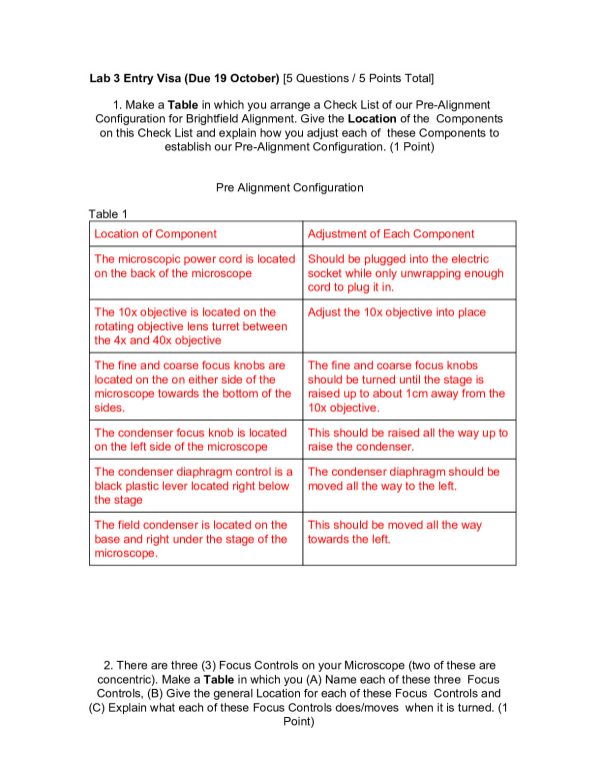
Molar Mass of a Volatile Liquid Lab
Group Members: Steven Soranno, Michael Pizzuto
Purpose: In this experiment the molar mass of an unknown volatile liquid will be determined by measuring what mass of vapor of the liquid is need to fill a flask of known volume at a particular temperature and pressure.
Materials:
· 250mLErlenmeyer Flask
· 1000mL Beaker
· Aluminum Foil
· Needle/pin
· Hot Plate
· Unknown liquid sample
· Ring Stand
· Thermometer
Procedure:
1. Prepare a 500-nt Erlenmeyer flask by cleaning the flask and then drying it completely.
2. The flask must be completely dry, since any water present will vaporize under the conditions of the experiment and will adversely affect the results. An oven may be available for heating the flask to dryness, or your instructor may describe another technique.
3. Cut a square of thick (freezer) aluminum foil to serve as a cover for the flask. Trim the edges of the foil so that it neatly covers the mouth of the flask but does not extend far down the neck.
4. Prepare a large beaker for use as a heating bath for the flask. The beaker must be large enough for most of the flask to be covered by boiling water when in the beaker. Add the required quantity of water to the beaker. Set up the beaker on a hot plate in the exhaust hood, but do not begin to heat the water bath yet.
5. Weigh the dry, empty flask with its foil cover to the nearest mg (0.001 g).
6. Obtain an unknown liquid and record its identification number.
7. Add 3—4 mL of liquid to the dry Erlenmeyer flask. Cover the flask with the foil cover, making sure that the foil cover is tightly crimped around the rim of the flask. Punch a single small hole in the foil cover with a needle or pin.
8. Heat the water in the beaker to boiling. When the water in the beaker begins to boil, adjust the temperature of the hotplate so that the water remains boiling but does not splash from the beaker.
9. Immerse the flask containing the unknown liquid in the boiling water so that most of the flask is covered with the water of the heating bath (see Figure 15-1). Clamp the neck of the flask to maintain the flask in the boiling water.
10. Watch the unknown liquid carefully. The liquid will begin to evaporate rapidly, and its volume will decrease. The amount of liquid placed in the flask is much more than will be necessary to fill the flask with vapor at the boiling water temperature. Excess vapor will be observed escaping through the pinhole made in the foil cover of the flask.
11. When it appears that all the unknown liquid has vaporized, and the flask is filled with vapor, continue to heat for 1—2 more minutes. Then remove the flask from the boiling water bath; use the clamp on the neck of the flask to protect your hands from the heat.
12. Set the flask on the lab bench, remove the clamp, and allow the flask to cool to room temperature. The liquid will reappear in the flask as the vapor in the flask cools. While the flask is cooling, measure and record the exact temperature of the boiling water in the beaker, as well as the barometric pressure in the laboratory.
13. When the flask has cooled completely to room temperature, carefully dry the outside of the flask to remove any droplets of water. Then weigh the flask, foil cover, and condensed vapor to the nearest mg (0.00 1 g).
14. Repeat the determination by adding another 3 -4-mL sample of unknown liquid. Reheat the flask until it is filled with vapor; cool, and the reweigh the flask. The weight of the flask after the second sample of unknown liquid is vaporized should agree with the first determination within 0.05 g. If it does not, do a third determination.
15. When two acceptable determinations of the weight of vapor needed to fill the flask have been obtained, remove the foil cover from the flask and clean it out.
16. Fill the flask to the very rim with tap water, cover with the foil cover, and weigh the flask, cover, and water to the nearest 0.1 g. Determine the temperature of the tap water in the flask. Using the density of water at the temperature of the water in the flask and the weight of water the flask contains, calculate the exact volume of the flask.
17. If no balance with the capacity to weigh the flask when filled with water is available, the volume of the flask may be approximated by pouring the water in the flask into a 1-L graduated cylinder and reading the water level in the cylinder.
18. Using the volume of the flask (in liters), the temperature of the boiling water bath (in kelvins), and the barometric pressure (in atmospheres), calculate the number of moles of vapor the flask is capable of containing. R = 0.0821 L atm/mol K.
19. Using the weight of unknown vapor contained in the flask, and the number of moles of vapor present, calculate the molar mass of the unknown liquid.
Pre-Laboratory Questions:
1. The method used in this experiment is sometimes called the vapor density method. Beginning with the ideal gas equation, show how the density of a vapor may be determined by this method.
2. 2.31 g of the vapor of a volatile liquid is able to fill a 498-mL flask at 100°C and 775 mm Hg, what is the molecular weight of the liquid? What is the density of the vapor under these conditions?
3. Why is a vapor unlikely to behave as an ideal gas near the temperature at which the vapor would liquefy?
.
Data Table:
Work for Data Table:
Questions:
1. Two methods were described for determining the volume of the flask used for the molar mass determination. Which method will give a more precise determination of the volume? Why?
2. It was important that the flask be completely dry before the unknown liquid was added so that water present would not vaporize when the flask was heated. A typical single drop of liquid water has a volume of approximately 0.05 mL. Assuming the density of liquid water is 1.0 g/ml, how many moles of water are in one drop of liquid, and what volume would this amount of water occupy when vaporized at 100°C and 1 atm?
Conclusion:
[Show Less]