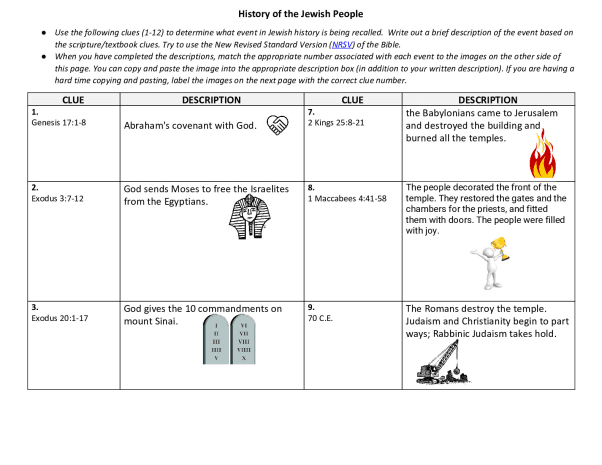
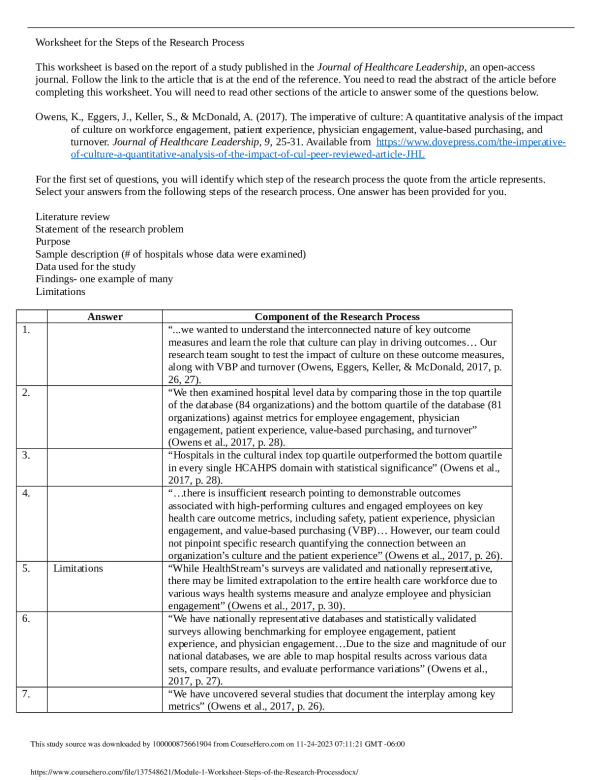
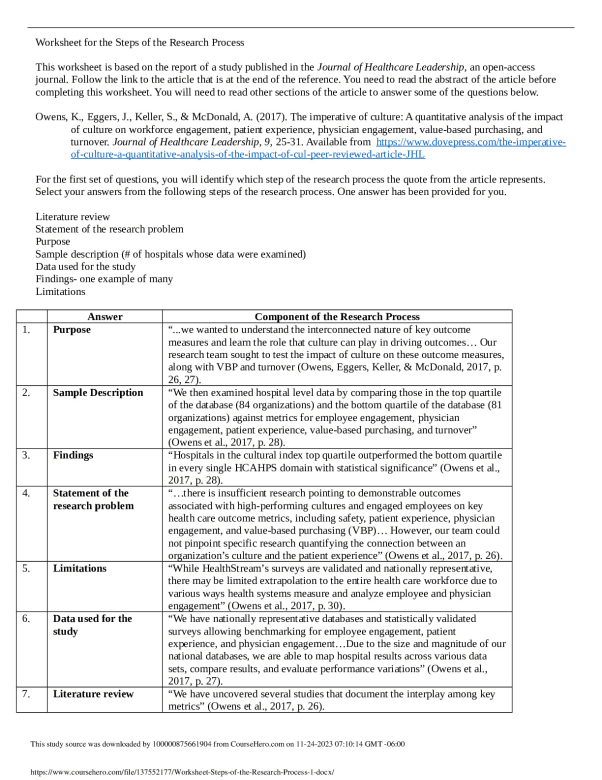
WORKSHEET > Worksheet 09 – Energy and Chemistry (Part 2)
University of British Columbia CHEM 154 Useful values: R = 8.3145 J mol-1 K-1 or 0.08206 L atm mol-1 K-1 0 1 2 3 4 5 6 7 8 9 Energy unit conversion: 1 L atm= 101.325 J 1) Calculate the work done (in J mol-1) for each of the following physical changes: a) The conversion of 1 mol of ice to 1 mol of water at 273 K and 1 atm. The molar volumes of ice an ...[Show More]
Preview 1 out of 4 pages
Reviews( 0 )
| Category: | |
| Number of pages: | 4 |
| Language: | English |
| Last updated: | 7 months ago |
| Downloads: | 1 |
| Views: | 0 |