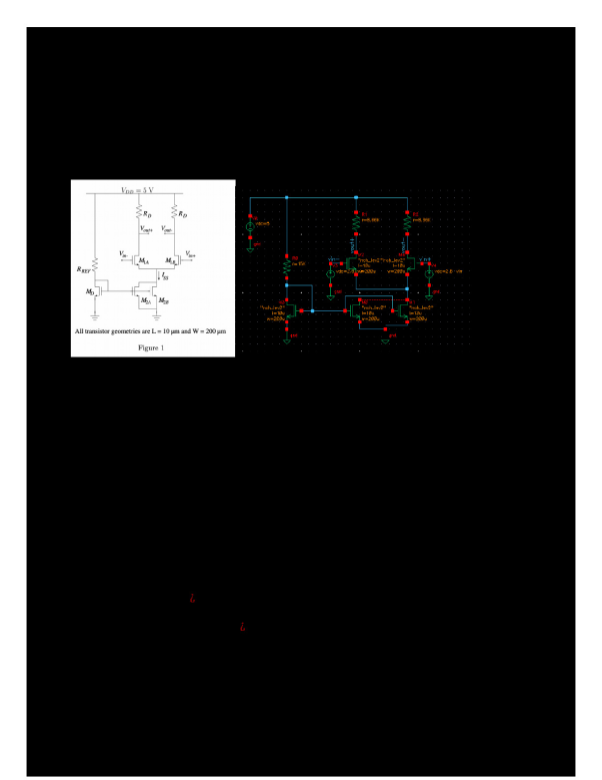
2.1.8 Lab 11: The Effects of Temperature and Particle Size Lab AssignmentAP Chemistry Sem 2 Name: ________________________Points Possible: 50 Date: _____________Adapted from: Kinetics Guided Inquiry, Chris Dewberry. https://phet.colorado.edu/en/contributions/view/4049DIRECTIONS:BackgroundFour major things affect the rate of a reaction:Concentration:More molecules = more collisionsCollisions are ne
...[Show More]
2.1.8 Lab 11: The Effects of Temperature and Particle Size Lab Assignment
AP Chemistry Sem 2 Name: ________________________
Points Possible: 50 Date: _____________
Adapted from: Kinetics Guided Inquiry, Chris Dewberry. https://phet.colorado.edu/en/contributions/view/4049
DIRECTIONS:
Background
Four major things affect the rate of a reaction:
Concentration:
More molecules = more collisions
Collisions are necessary for reactions
Physical State:
Ability to mix reactants
Solids can be grinded to increase surface area
Liquids can be stirred
Temperature:
Increases the kinetic energy of molecules
Need sufficient energy to react (Activation Energy)
Catalysts:
Typically lower the activation energy of reactions
Procedure
***For the majority of this guided inquiry we are going to use a simulation program written by
Linda Koch, Ron LeMaster, Trish Loeblein, and Kathy Perkins. The program can be found by
following this link: https://phet.colorado.edu/en/simulation/reactions-and-rates
Single Collision
1. Pull back the knob. Describe what happened in 2-3 sentences. (3 points)
2. Click the “Reload Launcher” button and expand the two windows on the right side of the
program by clicking the “+” button for the Separation View and the Energy View. Now
release the knob from various distances.
Indicate on the potential energy diagram when the reaction proceeds forward. (2 points)
When the total energy is equal or greater than the peak at which there is the greatest
amount of potential energy is when the reaction proceeds forward.
↓
This study source was downloaded by 100000784424693 from CourseHero.com on 05-30-2021 15:28:32 GMT -05:00
https://www.coursehero.com/file/91544249/218-Labdocxpdf/
This study resource was
shared via CourseHero.com
3. Now click the Angled Shot option in the top right corner. Try launching from a different
angle or two. Did the reaction proceed as before? (2 points)
The reaction did not proceed as before. Instead, the two reactants proceeded to bounce off of
eachother but not react.
4. Why do you think this was the outcome despite having enough energy for the reaction to
proceed? (3 points)
There was no direct collision of the two reactions. Because the two molecules did not collide
directly into each other, there was a low effectiveness of the collision.
5. Set the Choose a reaction option to the last preset chemical reaction that isn’t Design your
own. Now Change the Launcher Options back to Straight Shot and release the knob. What
happens to the translational speed of the molecules as the reaction goes forwards and
backwards? (Hint: the effect will be easiest to observe at a low energy). Please explain why
this occurs. (5 points)
The translational speed increases as the products are formed and slow down when the
reactants are formed. This is because there is more potential in the reactants than the
products as observed in the potential energy chart.
Rate Experiments
This study source was downloaded by 100000784424693 from CourseHero.com on 05-30-2021 15:28:32 GMT -05:00
https://www.coursehero.com/file/91544249/218-Labdocxpdf/
This study resource was
shared vi
[Show Less]






















-preview.png)