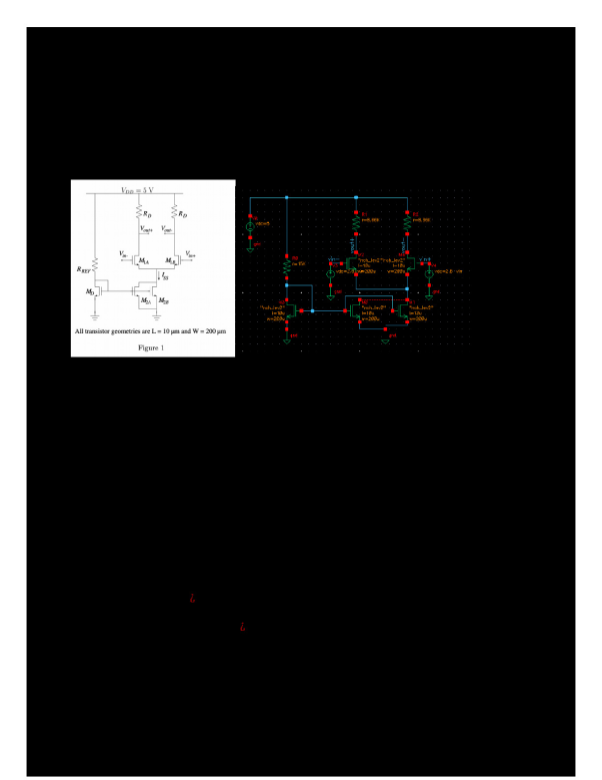
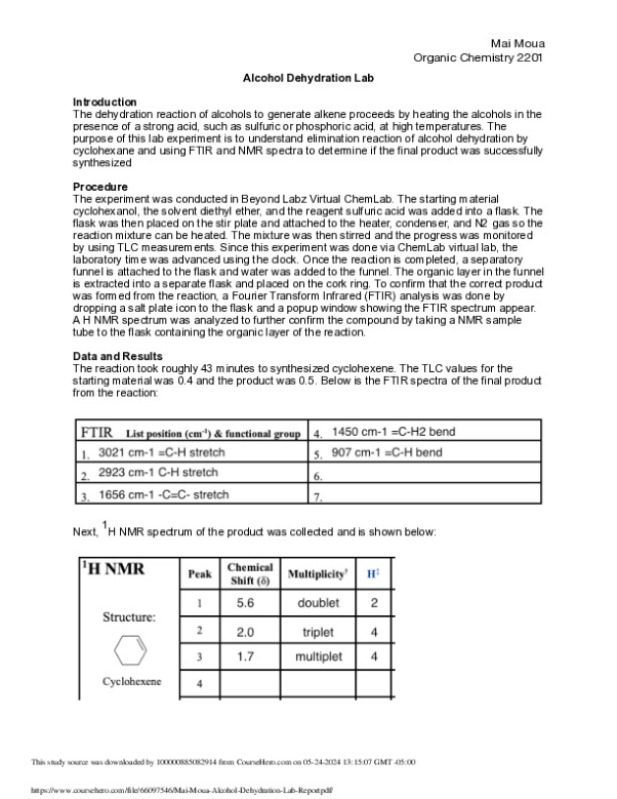
Chem 237L: Accurate and Precise Measurement of Volume
0 0 0 0 0.01 0.01 0.01 0.01 0.01 0.01 0.01
0
0.1
0.2
0.3
0.4
0.5
0.6
f(x) = 49.42x - 0.03
R² = 0.97
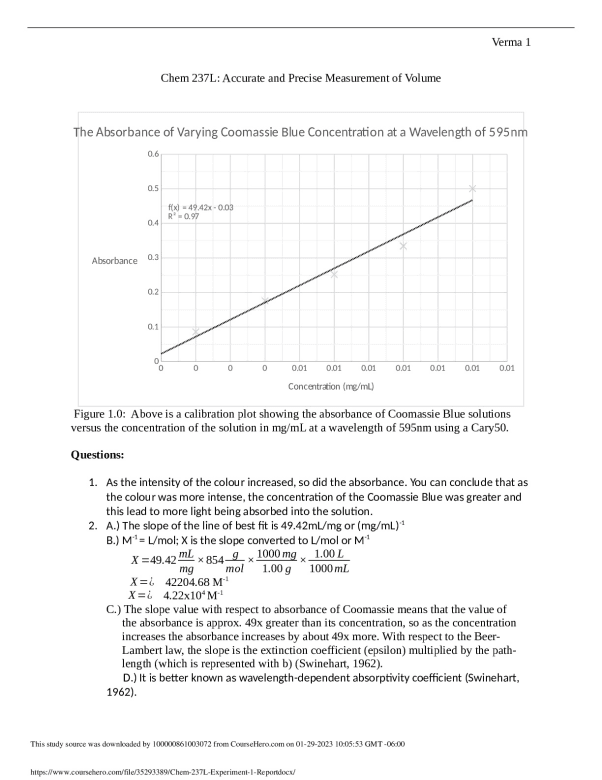
The Absorbance of Varying Coomassie Blue Concentration at a Wavelength of 595nm
Concentration (mg/mL)
Absorbance
Figure 1.0: Above is a calibration plot showing the absorbance of Coomassie Blue solutions
versus the concentration o
...[Show More]
Chem 237L: Accurate and Precise Measurement of Volume
0 0 0 0 0.01 0.01 0.01 0.01 0.01 0.01 0.01
0
0.1
0.2
0.3
0.4
0.5
0.6
f(x) = 49.42x - 0.03
R² = 0.97
The Absorbance of Varying Coomassie Blue Concentration at a Wavelength of 595nm
Concentration (mg/mL)
Absorbance
Figure 1.0: Above is a calibration plot showing the absorbance of Coomassie Blue solutions
versus the concentration of the solution in mg/mL at a wavelength of 595nm using a Cary50.
Questions:
1. As the intensity of the colour increased, so did the absorbance. You can conclude that as
the colour was more intense, the concentration of the Coomassie Blue was greater and
this lead to more light being absorbed into the solution.
2. A.) The slope of the line of best fit is 49.42mL/mg or (mg/mL)-1
B.) M-1 = L/mol; X is the slope converted to L/mol or M-1
X=49.42 mL
mg
× 854 g
mol ×
1000 mg
1.00 g
×
1.00 L
1000 mL
X=¿ 42204.68 M-1
X=¿ 4.22x104 M-1
C.) The slope value with respect to absorbance of Coomassie means that the value of
the absorbance is approx. 49x greater than its concentration, so as the concentration
increases the absorbance increases by about 49x more. With respect to the BeerLambert law, the slope is the extinction coefficient (epsilon) multiplied by the pathlength (which is represented with b) (Swinehart, 1962).
D.) It is better known as wavelength-d
[Show Less]





















-preview.png)