George Mason University
CHEM 214
G#: G01216043 CHEM 214-205
Experiment 007: Acidity Constant from a Titration Curve
References:
Suzanne Slayden, 2018, Chemistry 213, 214, 272 Laboratory Experiments, 7th Edition, pages 207-212.
George Mason University, Department of Chemistry and Biochemistry. Acidity Constant; 2020. Acidity Constant, Distance Learning Summer 20
...[Show More]
G#: G01216043 CHEM 214-205
Experiment 007: Acidity Constant from a Titration Curve
References:
Suzanne Slayden, 2018, Chemistry 213, 214, 272 Laboratory Experiments, 7th Edition, pages 207-212.
George Mason University, Department of Chemistry and Biochemistry. Acidity Constant; 2020. Acidity Constant, Distance Learning Summer 2020, accessed via Blackboard.
Purpose
The purpose of this experiment is to determine the acidity constant Ka of hydrochloric acid (HCl) and acetic acid (CH3COOH). This will be done by conducting two separate titrations of HCl and CH3COOH with a strong base (NaOH) and tracking the changing pH with a pH meter. The anticipated outcome is that the experimental Ka constants of HCl and CH3COOH will match the theoretical Ka constants of HCl and CH3COOH. The balanced equations for this experiment are:
HCl (aq) + NaOH (aq) → NaCl (aq) + H2O (l) CH3COOH (aq) + NaOH (aq) → CH3COONa (aq) + H2O (l)
Materials
• 25-mL Graduated Cylinder
• 50-mL Graduated Cylinder
• 45 mL of 0.1001-M NaOH Solution
• 10 mL of 0.1028-M HCl Solution
• 10 mL of 0.1020-M CH3COOH Solution
• pH Meter
• 50-mL Erlenmeyer Flask
• Phenolphthalein Indicator
• 2 Burets
• Distilled Water
Procedure
1. Titration of a Strong Acid with a Strong Base
a. Rise a clean buret with NaOH solution, then measure exactly 45 mL of NaOH with
a 50-mL graduated cylinder. Pour into buret.
b. Calibrate the pH meter.
c. Fill a 50-mL Erlenmeyer flask with exactly 10 mL of the HCl solution, then add a
couple drops of the phenolphthalein indicator. Record the pH of the HCl
solution.
d. Titrate HCl solution by adding the NaOH solution from the buret in 1.0 mL
sections.
e. Swirl flask after every addition of NaOH.
f. Remove the pH probe from the HCl solution, and then rinse it with distilled
water. Put probe back in the solution. Record pH of the HCl solution.
g. Start adding 0.2 mL sections instead of 1 mL sections of NaOH after 8 mL of
NaOH has been delivered.
h. Repeat Steps e and f.
i. Once the pH has a reading of 10 or more, stop adding NaOH in 0.2 mL sections.
Switch back to adding it in 1.0 mL sections.
j. Repeat Steps e and f.
k. When the pH does not change after 3 NaOH additions in a row, stop titration.
2. Titration of a Weak Acid with a Strong Base
a. Repeat Part 1 using CH3COOH instead of HCl.
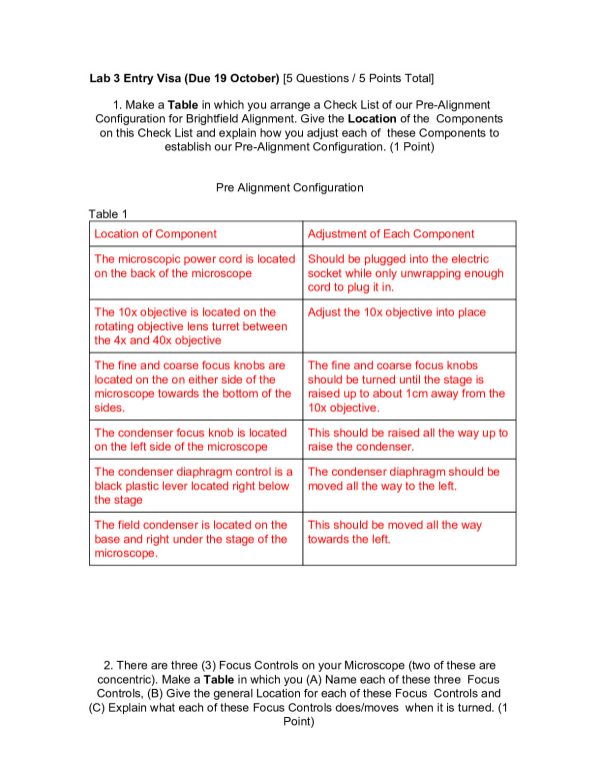
Data Table
Table 1: Raw Data for the Titration of HCl with NaOH. Christina Allen, G01216043, Chem-214-205
[Show Less]
-preview.png)