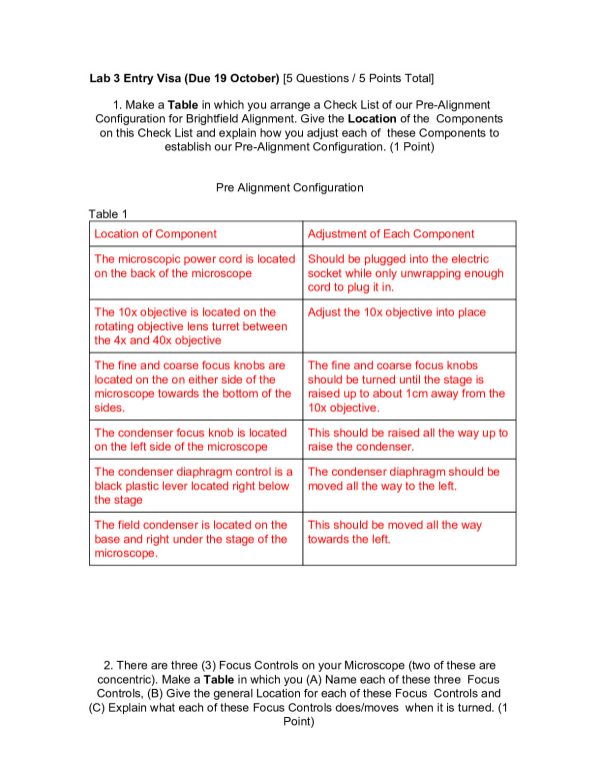
UNIVERSITY OF THE WITWATERSRAND
DEPARTMENT OF CHEMISTRY
CHEM3002/3003: PHYSICAL CHEMISTRY PRACTICAL COURSE
DETERMINATION OF THE SEDIMENTATION RATE AND PARTICEL
SIZE DISTRIBUTION FOR SILICA PARTICLES
1. INTRODUCTION
In this experiment, the distance with which silica particles travel through a water-Daxad 15 solution
within a given time will be measured. The resulting average velocity at whic
...[Show More]
UNIVERSITY OF THE WITWATERSRAND
DEPARTMENT OF CHEMISTRY
CHEM3002/3003: PHYSICAL CHEMISTRY PRACTICAL COURSE
DETERMINATION OF THE SEDIMENTATION RATE AND PARTICEL
SIZE DISTRIBUTION FOR SILICA PARTICLES
1. INTRODUCTION
In this experiment, the distance with which silica particles travel through a water-Daxad 15 solution
within a given time will be measured. The resulting average velocity at which the particles move
through the system can be determined with the measured parameters. The velocity of the particles
reveals information about size of the silica particles. Particle size analysis is a commonly used in
pharmaceutical industries for developing efficient asthma pumps, Ink related industries for ensuring
the correct viscosity and stability of manufactured inks as well as cement manufacturing to control
manufacturing costs by optimizing particles size (ATA Scientific Pty Ltd, 2018). The wide application
of this genre of analysis motivates this practical experiment. This experiment aims to educate the
experimenter on the practical workings of a telescope, to determine the size and size distribution for
silica particles in a silica suspension and to determine the radius of the most numerous particles in
the aforementioned distribution.
2. THEORETICAL BACKGROUND
In this experiment the rate of sedimentation for silica particles in a silica suspension will be
determined from two measured parameters; the length of sedimentation and the corresponding time
for the sedimentation to build. From basic mathematical analysis, the velocity can subsequently be
determined as seen in equation 1.
??
??
= ? (1)
In this experiment, it is assumed that the silica particles are spherical and consequently move with a
constant velocity. This assumption enables the application of Stoke’s Law. Stoke Law states that the
terminal settling velocity for sedimentation of spherical particles in a fluid is inversely proportional
to fluid’s viscosity and directly proportional to the difference in density of the particle and its
environment, the radius of the subject particles and the gravitational force the articles experience
(The Editors of Encyclopaedia Britannica, 2017). The law is summarized in equation 2, 3 and 4. It is
indeed correct to apply Stoke’s law I this experiment because the size of the subject particles are
small enough that their volume correctly approximated to a sphere
[Show Less]
-preview.png)