SCIENCE 2013400CA-7.04 HONORS CALORIMETRYHonors Calorimetry ActivityIn this activity, you will plan and conduct an investigation on energy transfers betweentwo liquids of different temperatures.Objective: Plan and then conduct an investigation on energy transfer between twoliquids of different temperatures.Materials: thermometer, two foam cups, measuring cup from your kitchen, hot water,and one li
...[Show More]
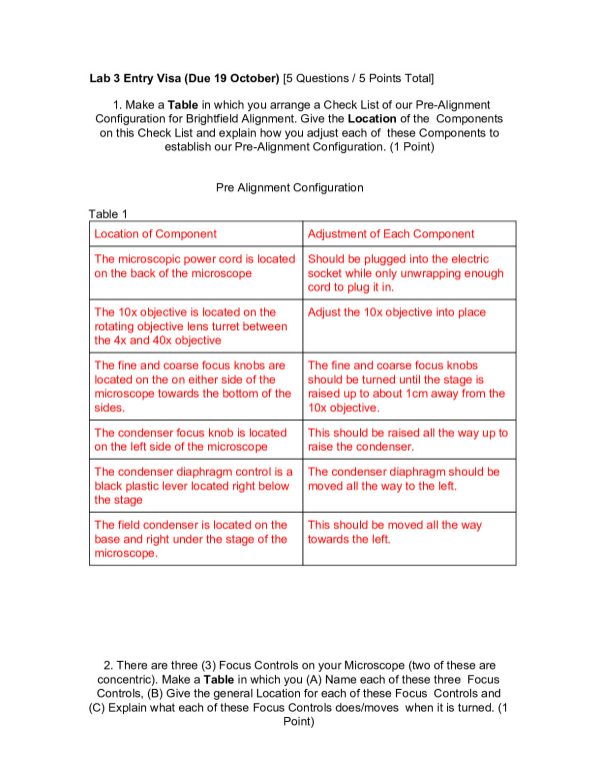
7.04 HONORS CALORIMETRY
Honors Calorimetry Activity
In this activity, you will plan and conduct an investigation on energy transfers between
two liquids of different temperatures.
Objective: Plan and then conduct an investigation on energy transfer between two
liquids of different temperatures.
Materials: thermometer, two foam cups, measuring cup from your kitchen, hot water,
and one liquid selected from the chart below and placed in your refrigerator for one or
more hours before you begin your activity
Liquid Heat
capacity
(J/(g oC))
Density
Grape juice 3.6 1.06 g/mL
Whole milk 3.77 1.04 g/mL
Orange
juice
3.73 1.05 g/mL
Tomato
juice
3.98 1.03 g/mL
Caution: You will be working with hot water in this lab. Be very careful as you pour so
you do not burn yourself. Also, be sure to read your thermometer carefully. The numbers
on the thermometer scale refer to the line under the number. Lastly, it's important to
perform certain steps quickly and efficiently to minimize heat loss to the surroundings.
Procedure:
1. Read the entire procedure before you begin the experiment.
2. Use the metric scale on your measuring cup to measure 237 mL (1 cup) of your
selected refrigerated liquid. Pour this liquid into one of the foam cups.
3. Now, get the hottest water possible from the faucet on your sink. Use the metric
scale on your measuring cup to measure 237 mL (1 cup) of hot water. Pour this
water into the other foam cup.
4. Quickly measure the temperature of the cold liquid and the hot water. Record the
temperatures in a data table.
[Show Less]