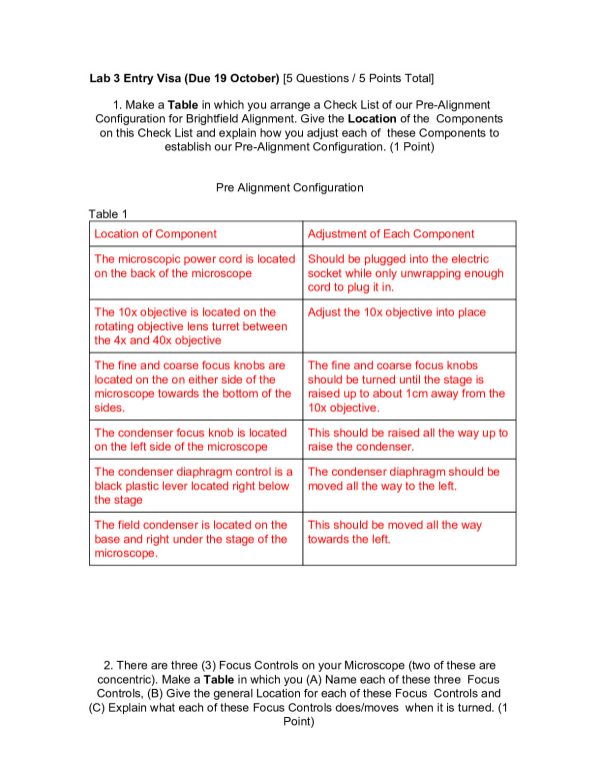
University of British ColumbiaCHEMISTRY 123Experiment 12 EDExperiment 12 – pH in BloodExperimental Design FormEDF 1Complete only page 1 and your procedural steps on page 2, BEFORE the start of your lab session.Include a screenshot of your Virtual Lab Feedback form in the Appendix on page 4.Research QuestionHow much Restoration liquid will be need to “save the patient” using a blood buffer sy
...[Show More]
Experiment 12 ED
Experiment 12 – pH in Blood
Experimental Design Form
EDF 1
Complete only page 1 and your procedural steps on page 2, BEFORE the start of your lab session.
Include a screenshot of your Virtual Lab Feedback form in the Appendix on page 4.
Research Question
How much Restoration liquid will be need to “save the patient” using a blood buffer system?
Hypothesis
25mL of Restoration liquid will be needed.
Glassware and equipment (include quantities)
Glassware / equipment Quantity Glassware / equipment Quantity
Beaker (250mL) 1 Graduated Cylinder 2
Volumetric Flask
(100mL)
1
Pasteur Pipet 1
pH Probe 1
Drop Dispenser 1
Pre-boiled Deionized
Water Bottle
1
Chemicals needed (names and amounts)
Calculations for the preparation of the buffer solution
(Can include as a screenshot in Appendix on page 4, if written by hand)
Procedural Steps, Observations and Changes to the Original Procedure
Chemical (name) Quantity
0.5M H2PO4 36.0mL
1.0M NaOH 12.0mL
0.1M HCl 29.30mL
0.1M NaOH 3.85mL
This study source was downloaded by 100000774734491 from CourseHero.com on 07-29-2022 14:58:05 GMT -05:00
https://www.coursehero.com/file/118379768/Experiment-12-EDFdocx/
E x p e r i m e n t 1 2 P a g e | 2
EDF 1 Procedural steps: Use 1 green row for each step. You can add more rows if needed but cannot
exceed this page.
EDF 2 Observations and Changes to original procedure: add these to the row below the procedural step
where the observation or change occurred. Remember to include reasons for changes.
Start by preparing 100mL of a phosphate buffer.
- Transfer 36mL of the 0.5M H2PO4 (phosphate buffer) into a clean 250mL beaker. To this
solution, add 1.0M NaOH while stirring until a pH of 7.45 is reached. Use a pH monitor to
monitor the pH. This buffer solution should now be transferred to a 100mL volumetric flask
and filled until the calibration point with pre-boiled deionized water. Add the formula to the
Microlab program and calibrate the drop dispenser.
- H2PO4 is a clear liquid.
- Put in an automatic stirrer into the beaker to mix the solution.
Titration
- Grab a 250mL beaker and pipet 25.00mL of the buffer solution. The drop dispenser should be
filled with 0.1M HCL and start the Microlab program. Dispense the HCl dropwise into the
buffer solution until the pH drops by 1 unit, then stop the program. This should give a.graph
which should be labelled HCl. Repeat the steps with 0.1M NaOH and title it NaOH. Mark the
graphs to display the acid and base buffer regions.
- HCl is a clear liquid.
- The HCl drops are slow and steady.
- The pH comes down slowly, it comes down by 0.1 by around every 2-3 drops of HCl.
- NaOH is a clear liquid.
- NaOH increases the pH very quickly, a small amount of NaOH was used.
[Show Less]