Stony Brook University
BIO 361
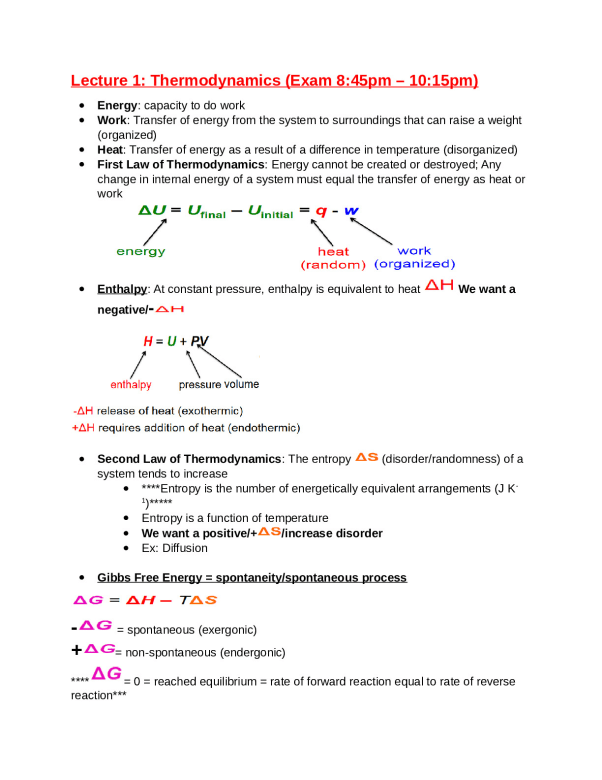
Lecture 1: Thermodynamics (Exam 8:45pm – 10:15pm) Energy: capacity to do work Work: Transfer of energy from the system to surroundings that can raise a weight (organized) Heat: Transfer of energy as a result of a difference in temperature (disorganized) First Law of Thermodynamics: Energy cannot be created or destroyed; Any change i
...[Show More]
Lecture 1: Thermodynamics (Exam 8:45pm – 10:15pm) Energy: capacity to do work Work: Transfer of energy from the system to surroundings that can raise a weight (organized) Heat: Transfer of energy as a result of a difference in temperature (disorganized) First Law of Thermodynamics: Energy cannot be created or destroyed; Any change in internal energy of a system must equal the transfer of energy as heat or work Enthalpy: At constant pressure, enthalpy is equivalent to heat We want a negative/- Second Law of Thermodynamics: The entropy (disorder/randomness) of a system tends to increase ****Entropy is the number of energetically equivalent arrangements (J K1 )***** Entropy is a function of temperature We want a positive/+ /increase disorder Ex: Diffusion Gibbs Free Energy = spontaneity/spontaneous process - = spontaneous (exergonic) + = non-spontaneous (endergonic) **** = 0 = reached equilibrium = rate of forward reaction equal to rate of reverse reaction*** Spontaneous at ALL temperatures: - and + = - = MOST IDEAL^^^ = exergonic R = gas constant / T = absolute temperature / Keq = equilibrium constant ***Standard Conditions: 25o C/1 atm/pH 7*** LARGE Keq = reaction tends to proceed until reactants have been almost completely converted into products Van’t Hoff Equation = measures equilibrium constant of a reaction at different temperatures - Left: high temp bottom/low temp top: Exothermic! = , and Y-intercept aka is less than 0/negative = spontaneous at LOW temperatures - Right: Endothermic = + Lecture 2: Waters and Buffers Hydrogen Bonds = ionic interaction, ***relatively weak at 20 kJ/mol***; they are longer and weaker than covalent bonds (below); however, bond energy HIGH due to numerous amounts of H2O molecules (which can form 4 hydrogen bonds = 80kj/mol) O-H bond = 470 kJ/mol C-C bond = 348 kJ/mol Water is a tetrahedral POLAR molecule; can donate two H’s and accept two H’s Oxygen partial charge = -0.66 e Hydrogen partial charge = +0.33 e Distance between two oxygens: 0.28 nm/2.8A Boiling point of water = 100oC vs methane = -164oC (similar structure but no H bonds) Networks of water molecules constantly breaking/re-forming every 2x10-11 seconds ***Many biological molecules can form hydrogen bonds including OH, C=O, carboxylate, ammonium**** Not all hydrogen bonds are the same Lone pair of electrons on oxygen pointing directly at hydrogen from donor molecule = strong H bond If at angle = weaker H bond Hydrophilic = tend
[Show Less]
-preview.png)
-preview.png)
-preview.png)





-preview.png)
