Massachusetts Institute of Technology
5. 12
Massachusetts Institute of Technology Organic Chemistry 5.12 March 8, 2019 Prof. Rick L. Danheiser Lecture 13: Electrophilic Addition to Alkenes III Lecture 13 - Electrophilic Addition to Alkenes III Learning Goals ① How to predict the products of reactions of alkenes with electrophilic reagents. ② Understanding the mechan
...[Show More]
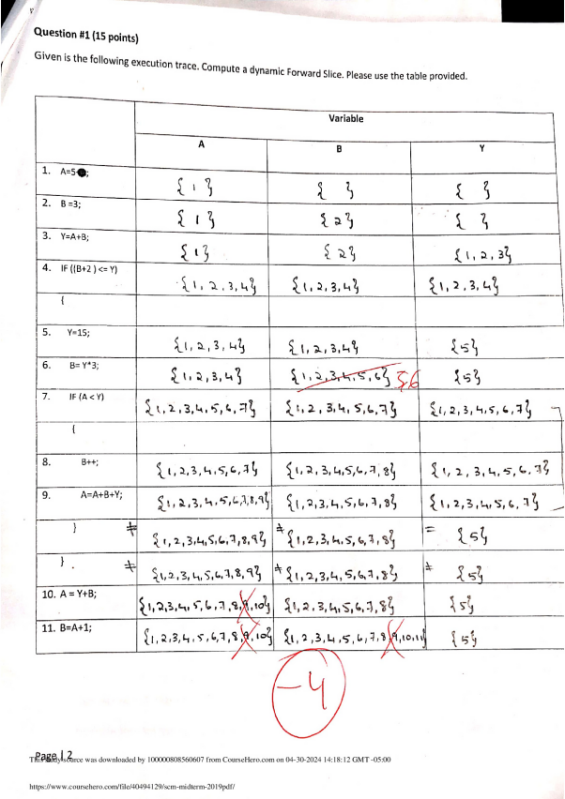
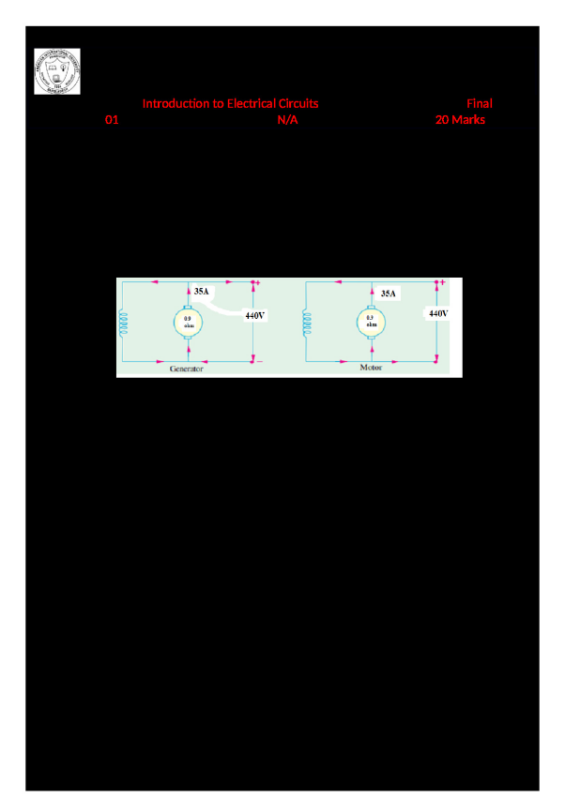
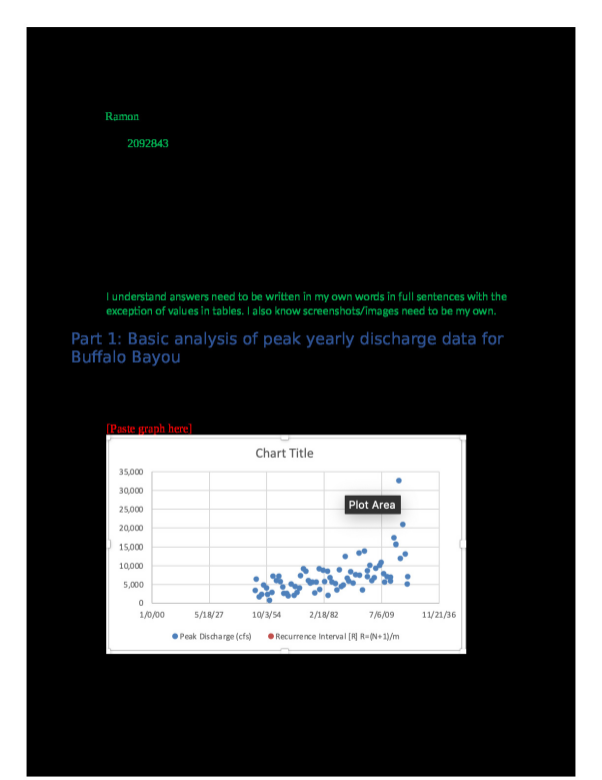
Massachusetts Institute of Technology Organic Chemistry 5.12 March 8, 2019 Prof. Rick L. Danheiser Lecture 13: Electrophilic Addition to Alkenes III Lecture 13 - Electrophilic Addition to Alkenes III Learning Goals ① How to predict the products of reactions of alkenes with electrophilic reagents. ② Understanding the mechanistic basis for the regiochemical and stereochemical outcome of these reactions Outline for Today's Lecture Halogenation of Alkenes Halohydroxylation of Alkenes Reading Assignment Lecture 13: Smith (5e & 4e) Chapter 10: 10.13-10.15 Lecture 14: Smith (5e & 4e) Chapter 10: 10.16-10.18; Chapter 12: 12.3-12.4 Practice Problems Smith (5e) Halogenation: 10.23, 10.24, 10.47 Halohydroxylation: 10.51 This study source was downloaded by 100000874968972 from CourseHero.com on 04-12-2024 11:25:32 GMT -05:00 This material may not be reproduced or posted on an internet website without written permission of Rick Danheiser and Christina Rotsides https://www.coursehero.com/file/47638769/512-Lecture13-3-08-19pdf/ ★ Alkenes have moderate nucleophilic character and eagerly react with strong electrophiles. ★ Hydrohalogenation involves addition of HCl, HBr, or HI to the double bond of alkenes. ★ Hydration adds HOH to alkenes and requires acid catalysis (e.g., with cat. H2SO4). ★ Hydrohalogenation and acid-catalyzed hydration proceed via carbocation intermediates which are powerful electrophiles. ★ Hydrohalogenation and acid-catalyzed hydration are regioselective processes that proceed via the formation of the more stable carbocation intermediate. ★ The Hammond Postulate tells us that the structure of the transition state for the formation of the carbocation resembles the carbocation and thus the more stable carbocation forms more rapidly (see Smith Sections 7.14 and 10.10). ★ Alkyl substituents are electron-donating groups and stabilize carbocations via hyperconjugation. ★ Since alkyl substituents are electron-donating groups, they increase the electron density in alkenes. ★ Hydrohalogenation and acid-catalyzed hydration usually are not stereoselective processes. The carbon atom in the carbocation intermediate is sp2 hybridized. Carbocations are planar and addition of nucleophiles often can
[Show Less]